(An Autonomous Body Recognized by Ministry of Commerce & Industry, Government of India)
Competency based placement focussed Education | Training | Research | Consultancy
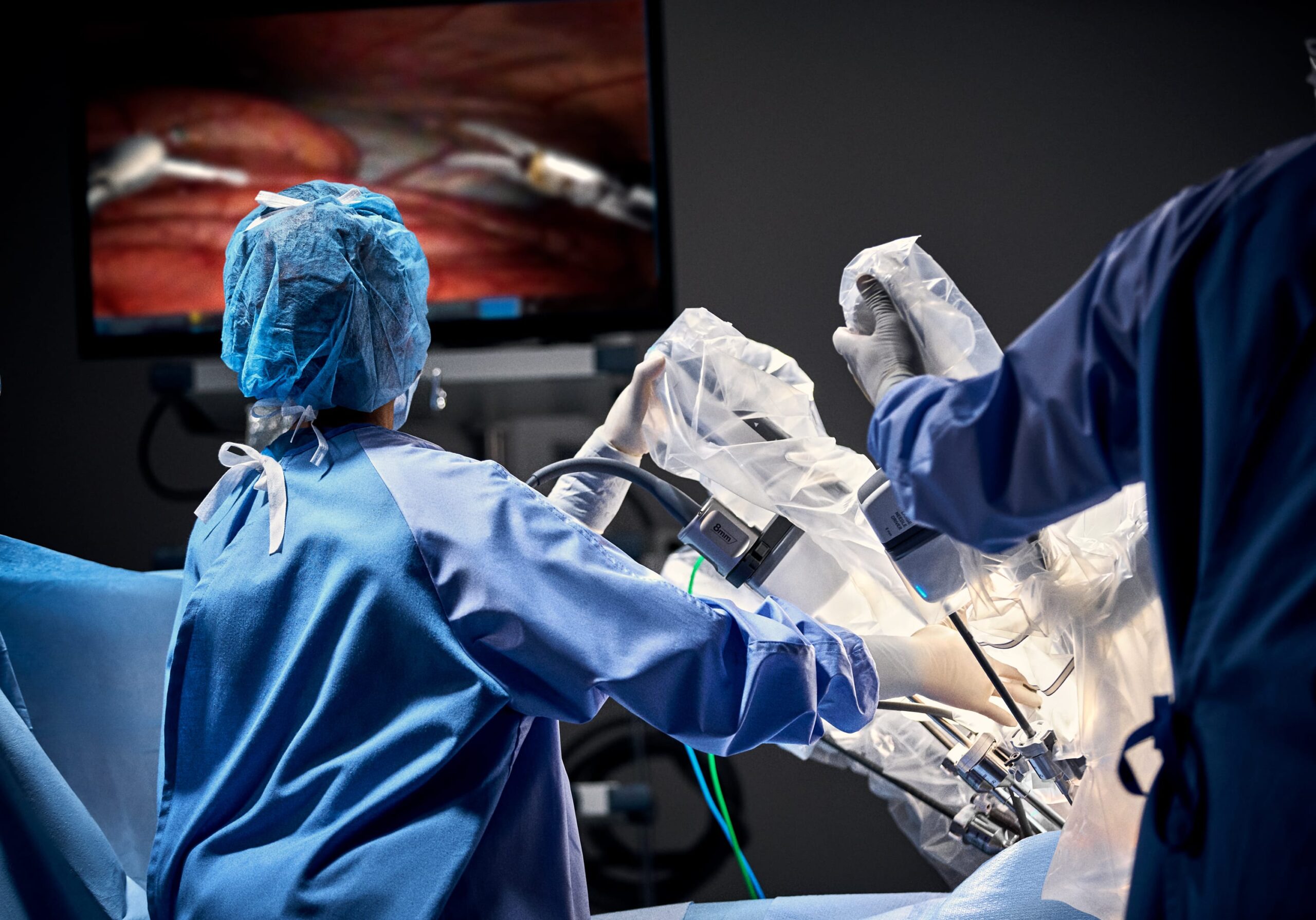
Restore Robotics Expands FDA-Cleared Portfolio for da Vinci Instruments
Restore Robotics has received additional 510(k) clearance from the Food and Drug Administration for two remanufactured instruments used with da Vinci Xi by Intuitive Surgical.
The newly cleared devices include the permanent cautery hook and cautery spatula, bringing Restore’s total FDA-cleared da Vinci instruments to four. Previous approvals covered monopolar curved scissors for both da Vinci Xi and Si systems.
The da Vinci Xi remains one of the most widely used robotic surgery platforms globally, with newer models like the da Vinci 5 driving continued growth.
Restore aims to offer hospitals cost-effective and sustainable alternatives by remanufacturing high-use surgical tools. The latest approvals strengthen efforts to reduce surgical costs without compromising performance.
The company plans to expand nationwide distribution through its partnership with Encore Medical Device Repair.
03-04-2026
📰 Recent News
- FDA Grants Breakthrough Device Status to Cognita’s Chest X-Ray AI
- Neuspera Reports Positive 12-Month Results for Urinary Incontinence Therapy
- FDA Grants De Novo Classification to LifeVac Anti-Choking Device
- FDA Clears Philips SmartHeart AI to Streamline Cardiac MR Imaging
- Francis Medical Launches Vanquish Prostate Ablation System in Arizona
- Polares Medical Secures $50m to Advance MRace Mitral Valve Implant
- FDA Grants Breakthrough Device Status to PathAI’s AI Dermatopathology Tool
- FDA Expands Clearance for Bayer’s Medrad MRXperion MR Injection System
- Philips Launches Rembra CT System for High-Demand Radiology Settings
- Airiver’s Drug-Coated Balloon Gains FDA Breakthrough Device Status
