(An Autonomous Body Recognized by Ministry of Commerce & Industry, Government of India)
Competency based placement focussed Education | Training | Research | Consultancy
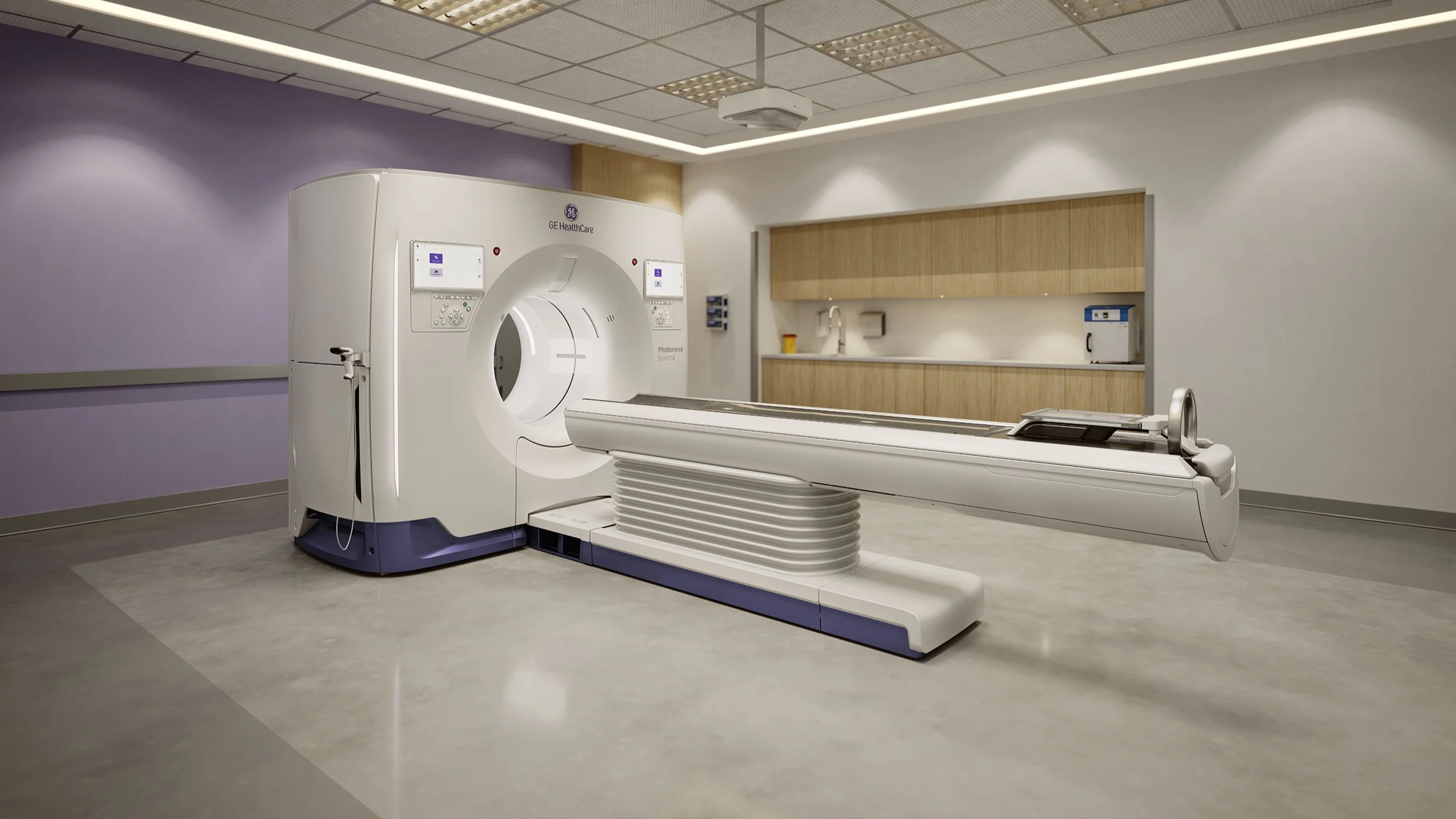
GE HealthCare Enters Photon-Counting CT Market with FDA Clearance
GE HealthCare has secured 510(k) clearance from the U.S. Food and Drug Administration for its Photonova Spectra system, marking its entry into the photon-counting CT segment. The approval positions the company to compete with Siemens Healthineers, whose Naeotom Alpha was the first such system cleared in the U.S.
Photon-counting CT technology improves imaging by directly measuring X-ray photons, eliminating the traditional light-conversion step and preserving more diagnostic data. GE’s system uses a “deep silicon” detector, designed to enhance contrast and enable clearer differentiation between materials such as calcium, iodine, and fat.
Analysts highlight its potential across multiple applications, including cardiac and neurological imaging, along with easier installation requirements that may speed adoption.
With orders expected to build through 2026, the technology could become a significant growth driver, with broader commercial impact anticipated in the following year.
23-03-2026
📰 Recent News
- FDA Grants Breakthrough Device Status to Cognita’s Chest X-Ray AI
- Neuspera Reports Positive 12-Month Results for Urinary Incontinence Therapy
- FDA Grants De Novo Classification to LifeVac Anti-Choking Device
- FDA Clears Philips SmartHeart AI to Streamline Cardiac MR Imaging
- Francis Medical Launches Vanquish Prostate Ablation System in Arizona
- Polares Medical Secures $50m to Advance MRace Mitral Valve Implant
- FDA Grants Breakthrough Device Status to PathAI’s AI Dermatopathology Tool
- FDA Expands Clearance for Bayer’s Medrad MRXperion MR Injection System
- Philips Launches Rembra CT System for High-Demand Radiology Settings
- Airiver’s Drug-Coated Balloon Gains FDA Breakthrough Device Status
