(An Autonomous Body Recognized by Ministry of Commerce & Industry, Government of India)
Competency based placement focussed Education | Training | Research | Consultancy
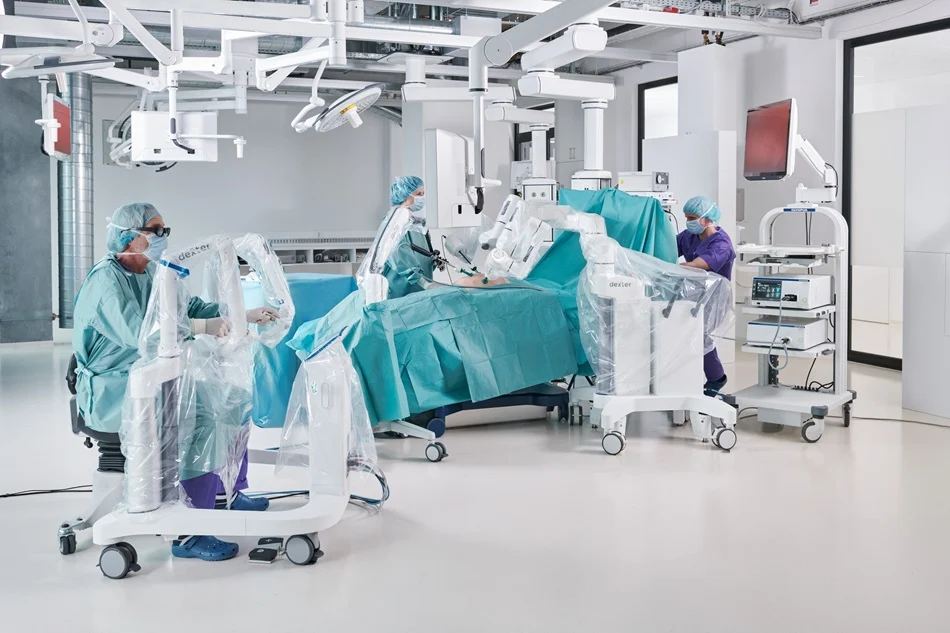
Distalmotion Seeks FDA Clearance to Expand Dexter Robot Use in Gynecology
Distalmotion has submitted a 510(k) application to the Food and Drug Administration to expand indications for its Dexter robotic surgery system in the U.S.
The company aims to extend Dexter’s use into gynecological procedures such as sacrocolpopexy, sacrocervicopexy, and endometriosis resection. The move supports its strategy to target ambulatory surgical centers (ASCs), where space and infrastructure constraints may favour compact robotic systems.
Dexter is already cleared for procedures including gallbladder removal, hernia repair, and hysterectomy. Expanding into outpatient gynecology marks a key step in the platform’s evolution.
Distalmotion recently completed a safety study in pelvic procedures and secured approval to begin trials in myomectomy. The company is positioning Dexter as a flexible, mobile solution to capture growing demand for robotic-assisted outpatient surgeries.
06-04-2026
📰 Recent News
- ARPA-H Launches ‘Delphi’ Program to Advance Next-Gen Biosensor Wearables
- Vitestro Raises $70M to Advance Autonomous Blood Collection Robot
- Insulet Advances Fully Closed-Loop System for Type 2 Diabetes
- FDA Warning Letter Highlights Quality Gaps at Advita Ortho Facility
- Insulet Recalls Omnipod 5 Pumps Over Insulin Delivery Issue
- FDA Grants Breakthrough Device Status to Cognita’s Chest X-Ray AI
- Neuspera Reports Positive 12-Month Results for Urinary Incontinence Therapy
- FDA Grants De Novo Classification to LifeVac Anti-Choking Device
- FDA Clears Philips SmartHeart AI to Streamline Cardiac MR Imaging
- Francis Medical Launches Vanquish Prostate Ablation System in Arizona
